Chapter : 3. Atoms & Molecules
Mole Concept
Atoms and molecules are so small in size that they cannot be counted individually. The chemists use the unit mole for counting atoms, molecules or ions. It is represented by n. A mole represents 6.022 × 1023 particles.
Example. 1 mole of atoms = 6.022 × 1023 atoms.
1 mole of molecules = 6.022 × 1023 molecules The number of particles present in 1 mole of any substance is fixed i.e. 6.022 × 1023.
This number is called Avogadro constant or Avogadro number. It is represented by No. 1 mole of atoms = 6.022 × 1023 atoms = Gram atomic mass or Molar mass of element
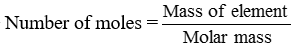

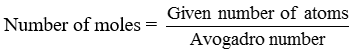
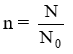
No. of moles = n
Given mass = m
Molar mass = M
Given number of particles = N
Avogadro number of particles = N0
These relations can be interchanged as
Mass of element, m = n × M
or No. of particles of element, N = n × N0
Similarly,
1 Mole of molecules = 6.022 × 1023 molecules
= Gram molecular mass of Molar mass


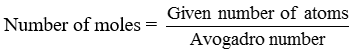
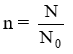
or m = n × M and N = n × N0
Example. 1 mole of atoms = 6.022 × 1023 atoms.
1 mole of molecules = 6.022 × 1023 molecules The number of particles present in 1 mole of any substance is fixed i.e. 6.022 × 1023.
This number is called Avogadro constant or Avogadro number. It is represented by No. 1 mole of atoms = 6.022 × 1023 atoms = Gram atomic mass or Molar mass of element
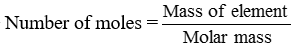

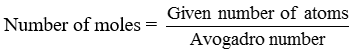
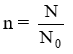
No. of moles = n
Given mass = m
Molar mass = M
Given number of particles = N
Avogadro number of particles = N0
These relations can be interchanged as
Mass of element, m = n × M
or No. of particles of element, N = n × N0
Similarly,
1 Mole of molecules = 6.022 × 1023 molecules
= Gram molecular mass of Molar mass


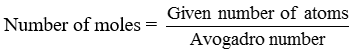
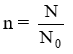
or m = n × M and N = n × N0
Trending Articles & Blogs
- Physics Tutor, Math Tutor Improve Your Child’s Knowledge
- How to Get Maximum Marks in Examination Preparation Strategy by Dr. Mukesh Shrimali
- 5 Important Tips To Personal Development Apply In Your Daily Life
- Breaking the Barriers Between High School and Higher Education
- 14 Vocational courses after class 12th
- Tips to Get Maximum Marks in Physics Examination
- Get Full Marks in Biology Class 12 CBSE
Download Old Sample Papers For Class X & XII
Download Practical Solutions of Chemistry and Physics for Class 12 with Solutions
Recent Questions Asked
- Newton’s laws of motion asked by Dr. Mukesh Shrimali
- Process of nutrition in Amoeba asked by Rajiv Sharma
- Importance of studying physics subject in school after 10th asked by Rajiv
- Refraction Through Prism in Different Medium asked by Kirti Sharma
- Ratio and Proportion Question asked by Education Desk
- Explain all the 12 tenses with example asked by Qwerty
- Refraction Through Prism in Different Medium asked by Seema Shrimali