Chapter : 1. Metals & Non Metals
Occurrence of metals in Nature
Occurrence Of Metals In Nature
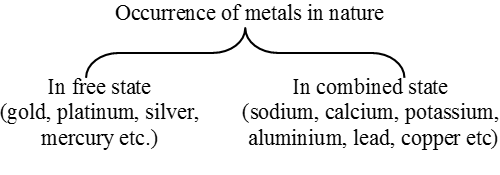
Metals occur in nature in free state or in combined state. A metal is said to occur native or free when it is found in nature in the metallic state. For example, gold may be found in nature as metal. This is because gold when left exposed to air practically does not undergo any change. It is not reacted upon by moisture, oxygen and carbon dioxide of the air. Thus, those metals which remain unaffected by moisture, oxygen and carbon dioxide of the air can occur native or free. In other words, the unreactive metals occur in nature in free state because of their low reactivity towards chemical reagents. Another example of an unreactive metal is silver.
The reactive metals, i.e., the metals which react with moisture, oxygen, carbon dioxide or other chemical reagents, are not found in nature in free state, but in combined state in the form of compounds.
Metals usually occur in combination with nonmetallic elements. The native occurrence is comparatively rare.
Metals occur in nature in free state or in combined state. A metal is said to occur native or free when it is found in nature in the metallic state. For example, gold may be found in nature as metal. This is because gold when left exposed to air practically does not undergo any change. It is not reacted upon by moisture, oxygen and carbon dioxide of the air. Thus, those metals which remain unaffected by moisture, oxygen and carbon dioxide of the air can occur native or free. In other words, the unreactive metals occur in nature in free state because of their low reactivity towards chemical reagents. Another example of an unreactive metal is silver.
The reactive metals, i.e., the metals which react with moisture, oxygen, carbon dioxide or other chemical reagents, are not found in nature in free state, but in combined state in the form of compounds.
Metals usually occur in combination with nonmetallic elements. The native occurrence is comparatively rare.

Trending Articles & Blogs
- Physics Tutor, Math Tutor Improve Your Child’s Knowledge
- How to Get Maximum Marks in Examination Preparation Strategy by Dr. Mukesh Shrimali
- 5 Important Tips To Personal Development Apply In Your Daily Life
- Breaking the Barriers Between High School and Higher Education
- 14 Vocational courses after class 12th
- Tips to Get Maximum Marks in Physics Examination
- Get Full Marks in Biology Class 12 CBSE
Download Old Sample Papers For Class X & XII
Download Practical Solutions of Chemistry and Physics for Class 12 with Solutions
Recent Questions Asked
- Newton’s laws of motion asked by Dr. Mukesh Shrimali
- Process of nutrition in Amoeba asked by Rajiv Sharma
- Importance of studying physics subject in school after 10th asked by Rajiv
- Refraction Through Prism in Different Medium asked by Kirti Sharma
- Ratio and Proportion Question asked by Education Desk
- Explain all the 12 tenses with example asked by Qwerty
- Refraction Through Prism in Different Medium asked by Seema Shrimali